Helping You Stay
COMPETITIVE, Compliant,
EFFICIENT &
Profitable
Your project has unique needs and requirements. We put in the work to understand how to serve you best, so you get the right solution and achieve your project’s goals.
Get in Touch
we are

(in-teh-gray-o)
Serving Companies with Critical Facilities in the
Pharmaceutical, Biotech, Semiconductor and Data Center Sectors
Commissioning & Qualification
Successful commissioning and qualification requires a thorough understanding of engineering, quality assurance, manufacturing, and regulatory compliance. To ensure your project goals are met, we develop and execute detailed processes and focus on maintaining clear and consistent communication throughout every phase of the project.
Learn More
MEP
ENGINEERING
Whether you have a new project or a renovation, our MEP engineers focus on designing systems that ensure you achieve a high-quality and timely end result.
Learn More

We Understand
Solutions That Provide Tangible ValueAs a manufacturing business, you constantly grapple with compliance, operating, equipment, and facilities challenges. To stay competitive and profitable, you need to manage your costs while not sacrificing your product’s quality. We work with you to provide solutions that have a clear return on investment and deliver real value to your operators and management teams.
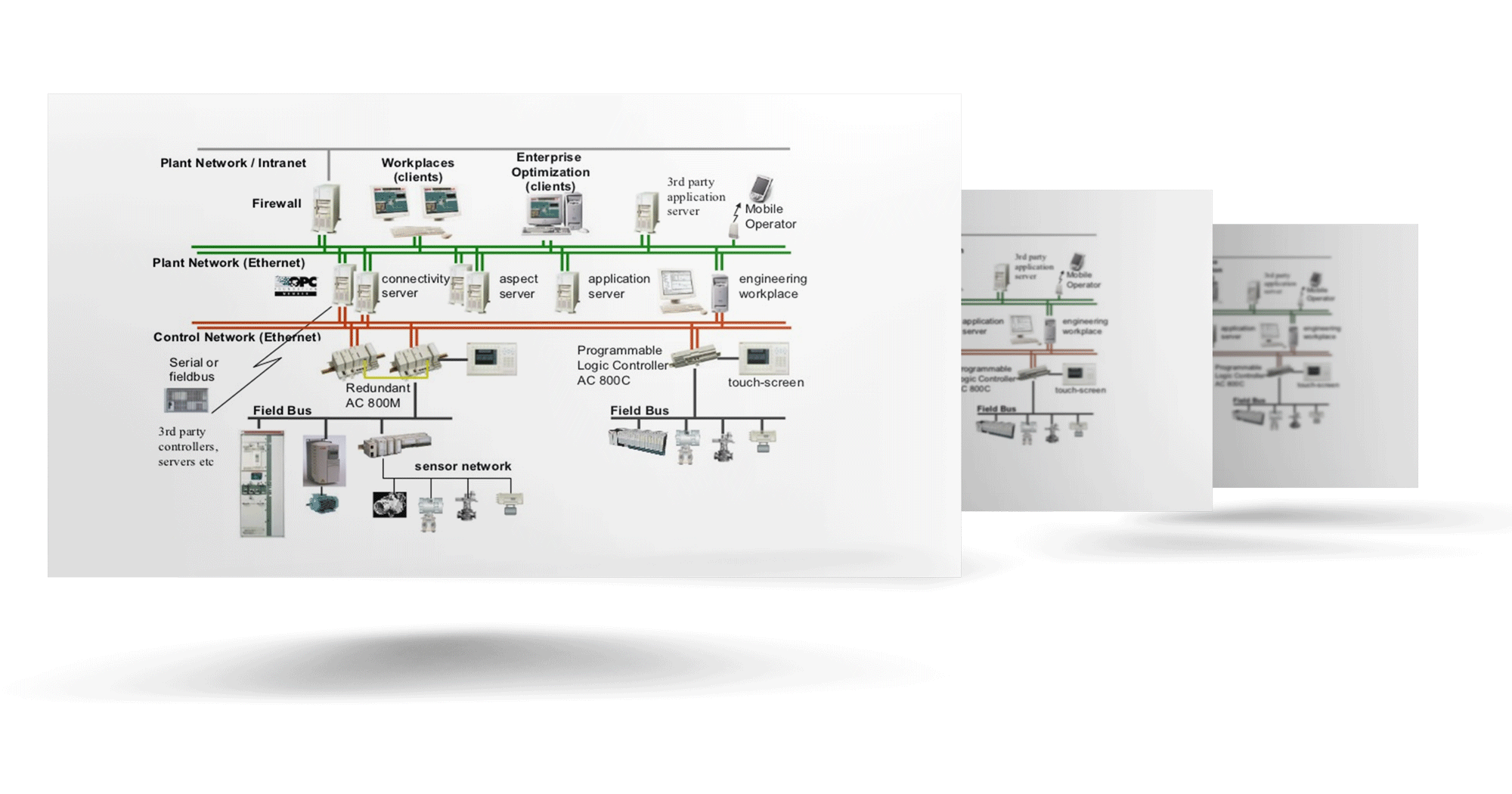
INDUSTRIALAUTOMATION
Automation must provide substantial business value. We measure that value based on enhanced asset availability, return on assets, reduced lifecycle cost, and higher quality and volume of products produced. Integrao provides automation solutions that deliver on each of these metrics through industry-leading hardware and software.
Learn More
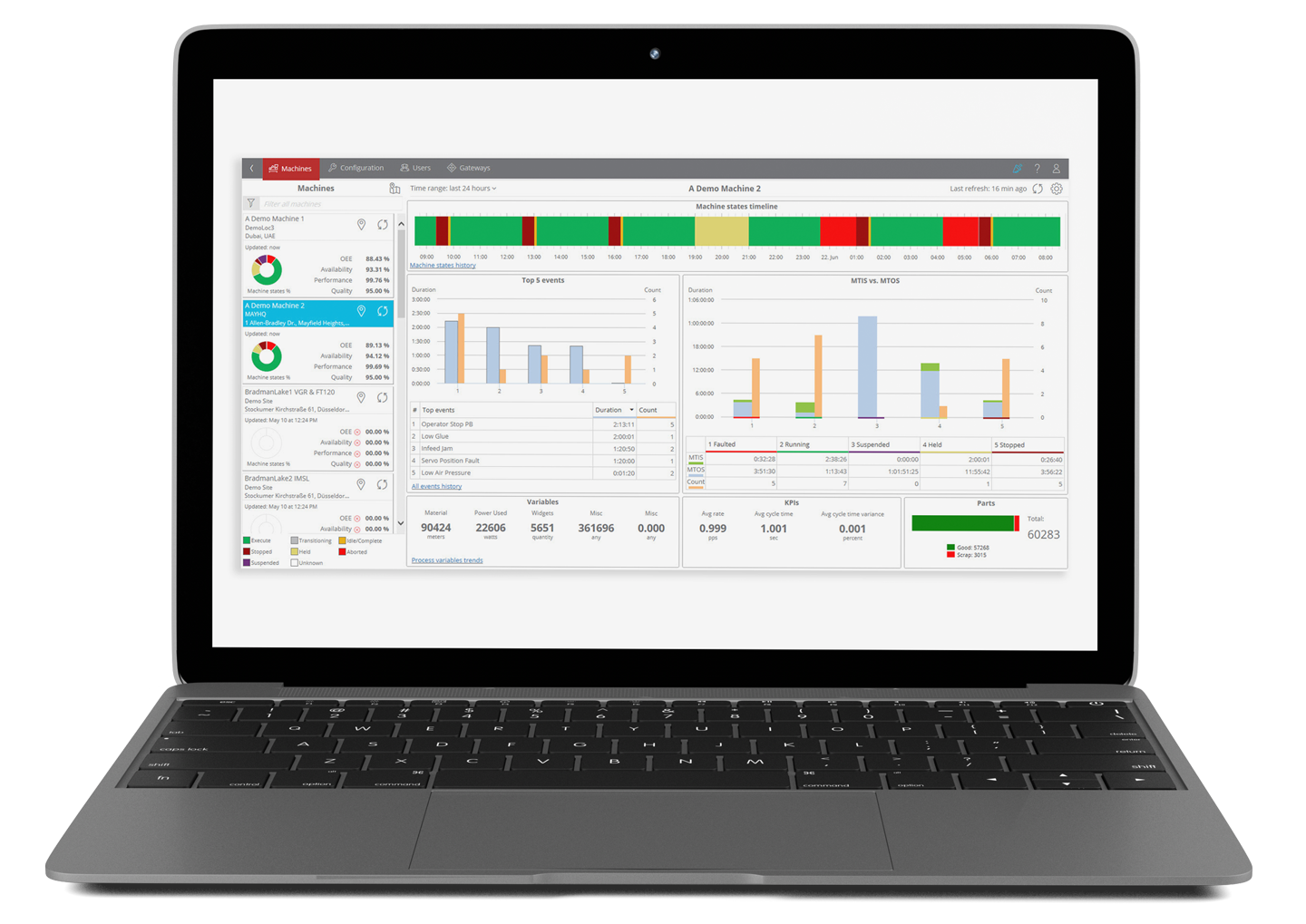
OperationalAnalytics
Integrao team members will help your organization focus on expected outcomes for each unit operation process. We help each customer make sense of the data generated by their processes and then work with them to determine how to use it to modify or enhance their systems. We can provide this information to all members of your team, company, or the entire value chain.
compliance
21 CFR Part 211 and ASHRAE 0-2019To achieve regulatory compliance, pharmaceutical and biotechnology manufacturing facilities need good documentation. The Integrao team works with your cross-functional teams to ensure you can leverage all building and process systems testing documentation for the validation process. We generate test protocols, perform tests, and commission systems according to industry standards such as ASHRAE Guideline 0-2019, ASTM E2500-13, and FDA Pharmaceutical cGMPs for the 21st Century.
Learn More



